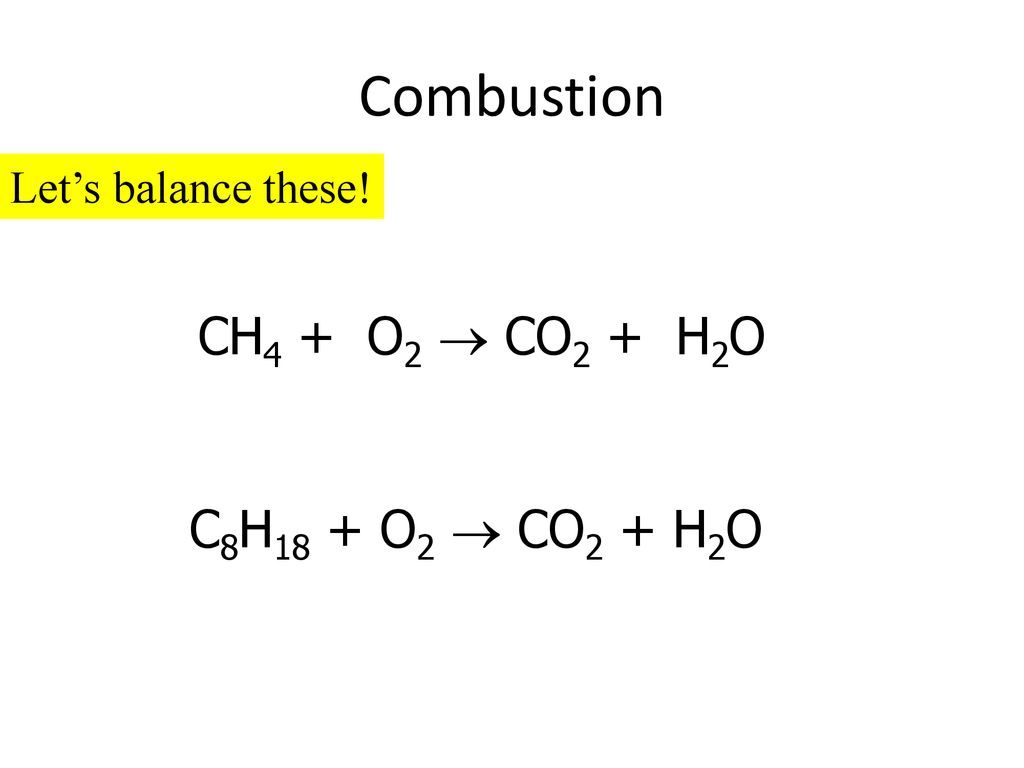
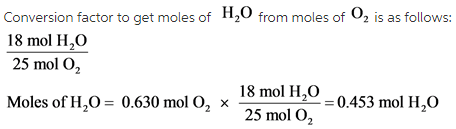How to C8H18+O2=CO2+H2O|Chemical equation C8H18+O2=CO2+H2O|Reaction balance C8H18+O2=CO2+H2O| - YouTube
C8H18 +O2 =CO2 + H2O Balance ||Octane Complete Combustion Balanced Equation| C8H8 +O2 YieldsCO2,H2O دیدئو dideo

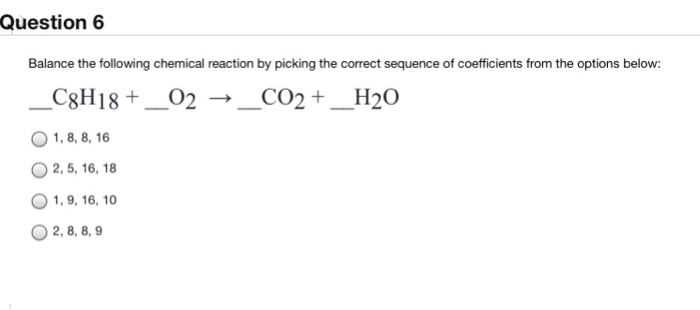
SOLVED: SECTION 3: ANSWER ONE QUESTION FROM THIS SECTION. Balance the following chemical reactions. (2 Marks Each) C8H18 + O2 → CO2 + H2O C9H20 + O2 → CO2 + H2O +
When 10 g of CxHy is burned, 45.00 g of products are formed (CO2 and H2O combined), what is the empirical formula of the fuel (CxHy + O2→ CO2 + H2O)? - Quora

C8H18 +O2 =CO2 + H2O Balance ||Octane Complete Combustion Balanced Equation| C8H8 +O2 YieldsCO2,H2O - YouTube