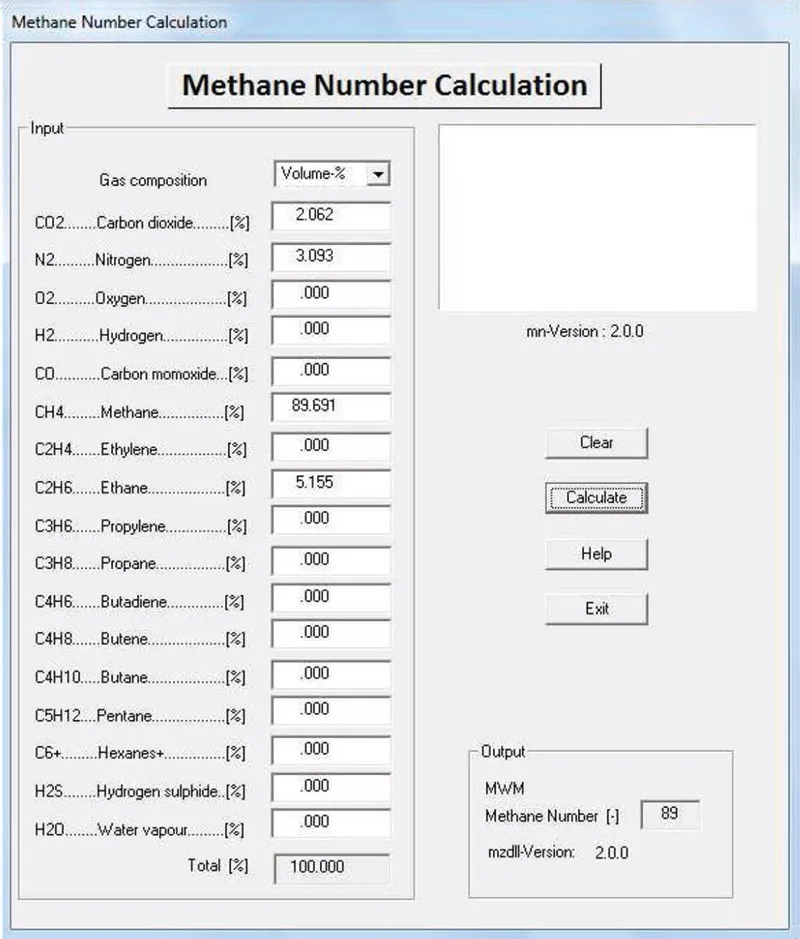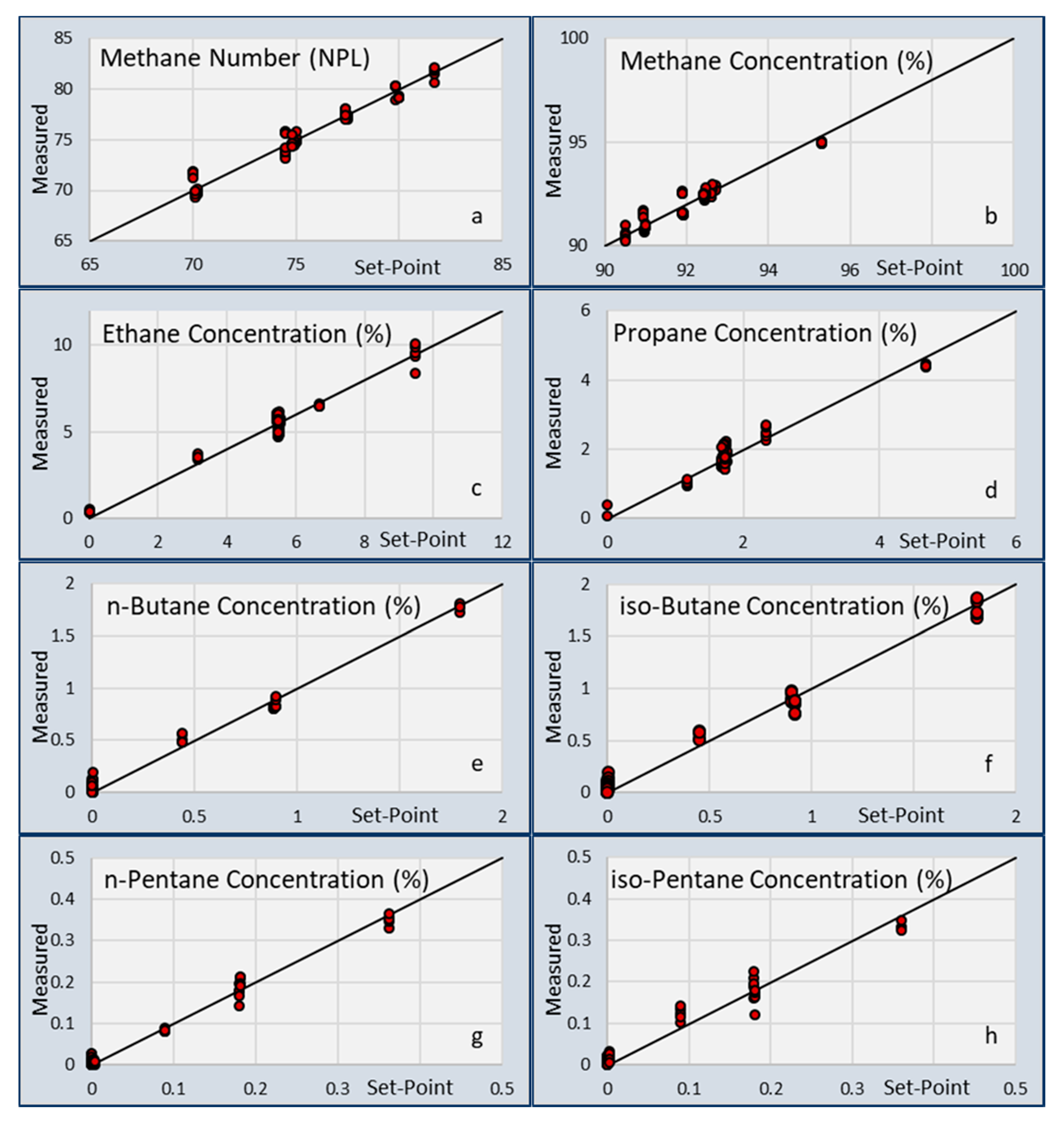
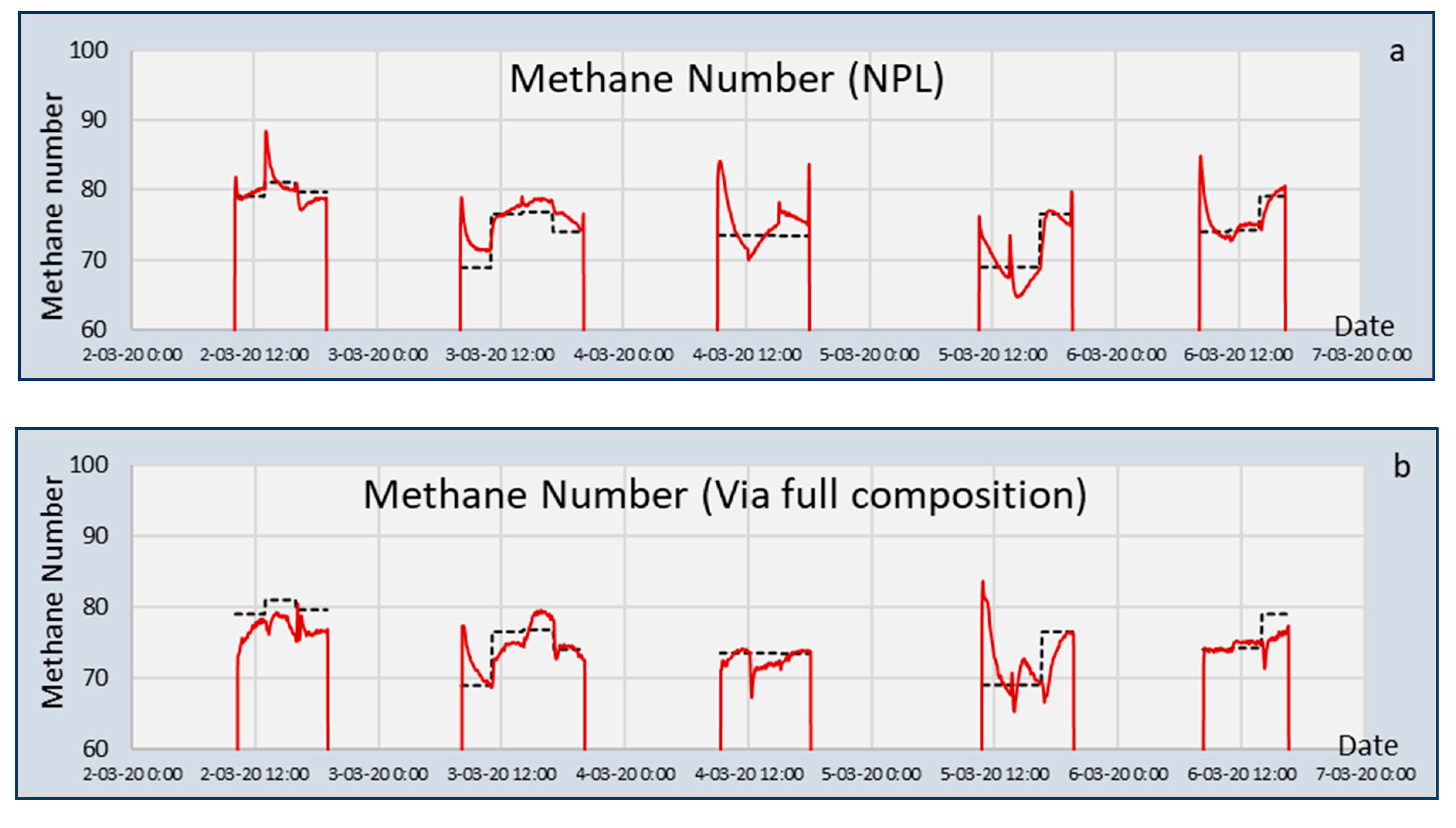
Sensors | Free Full-Text | Capacitive and Infrared Gas Sensors for the Assessment of the Methane Number of LNG Fuels

PROBLEM 1.3 Calculate the number of moles and molecules present in 0.032 mg of methane (C = 12, H =1) Solution : Molar mass of methane, CH, = 12 + 4 =
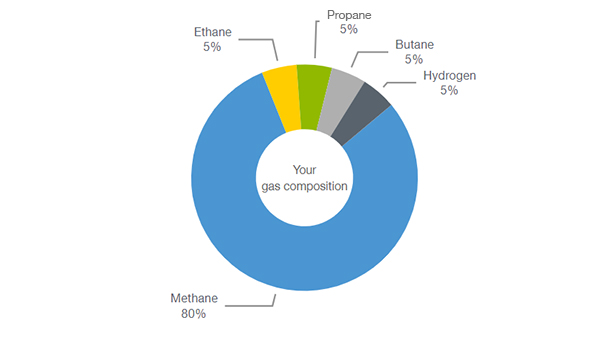









![Methane [CH4] Molecular Weight Calculation - Laboratory Notes Methane [CH4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/10/methane-molecular-weight-calculation-300x187.jpg)